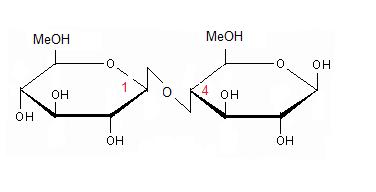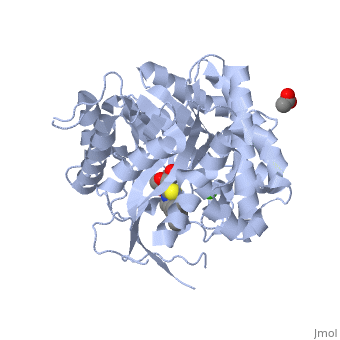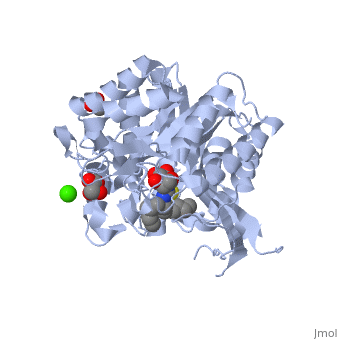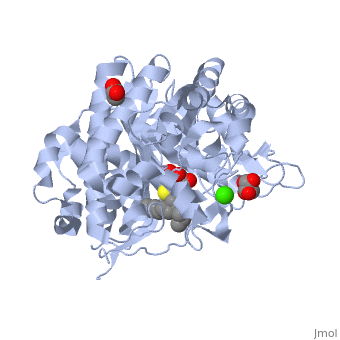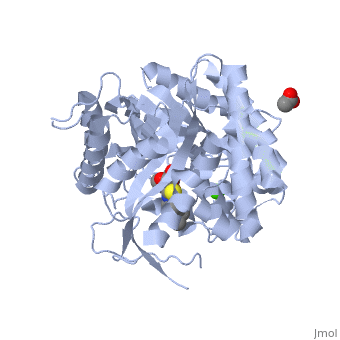
An engineered GH1 β-glucosidase displays enhanced glucose tolerance and increased sugar release from lignocellulosic materials | Scientific Reports
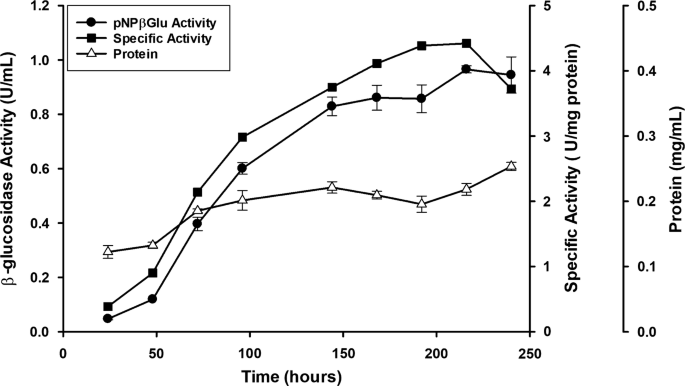
Penicillium citrinum UFV1 β -glucosidases: purification, characterization, and application for biomass saccharification | Biotechnology for Biofuels | Full Text

A promiscuous beta-glucosidase is involved in benzoxazinoid deglycosylation in Lamium galeobdolon - ScienceDirect
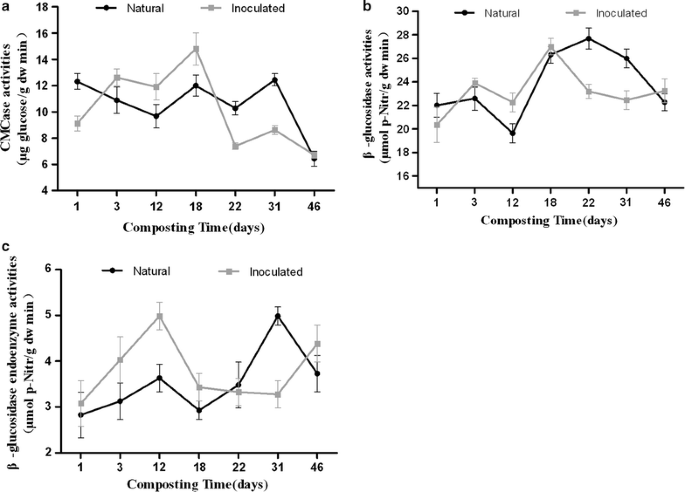
The structural and functional contributions of β-glucosidase-producing microbial communities to cellulose degradation in composting | Biotechnology for Biofuels | Full Text

The mechanism of substrate (aglycone) specificity in β-glucosidases is revealed by crystal structures of mutant maize β-glucosidase-DIMBOA, -DIMBOAGlc, and -dhurrin complexes | PNAS

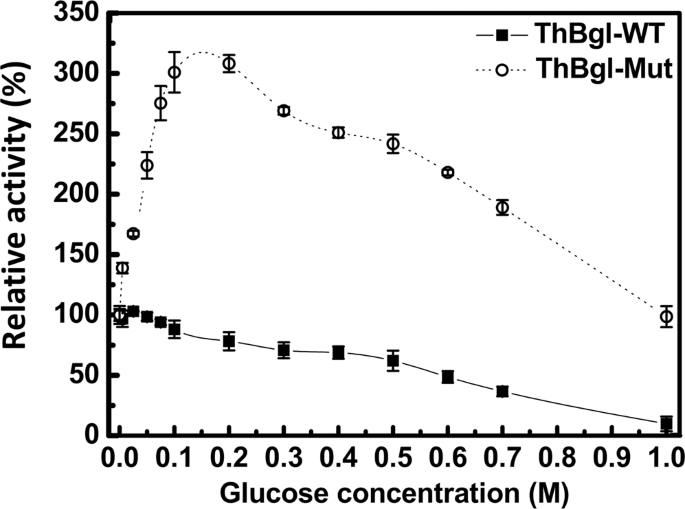
Overexpression and characterization of a glucose-tolerant β-glucosidase from Thermotoga thermarum DSM 5069T with high catalytic efficiency of ginsenoside Rb1 to Rd - ScienceDirect
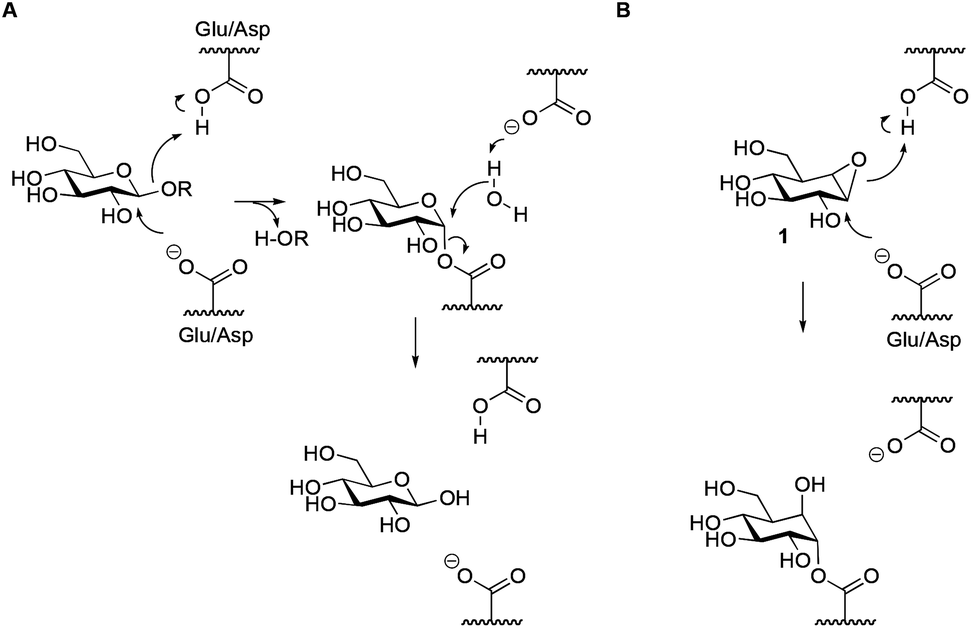
Exploring functional cyclophellitol analogues as human retaining beta- glucosidase inhibitors - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C4OB01611D

A novel strategy for efficient disaccharides synthesis from glucose by β - glucosidase | SpringerLink
Engineering the GH1 β-glucosidase from Humicola insolens: Insights on the stimulation of activity by glucose and xylose

The carbon source determines the nature of the b-glucosidase produced.... | Download Scientific Diagram
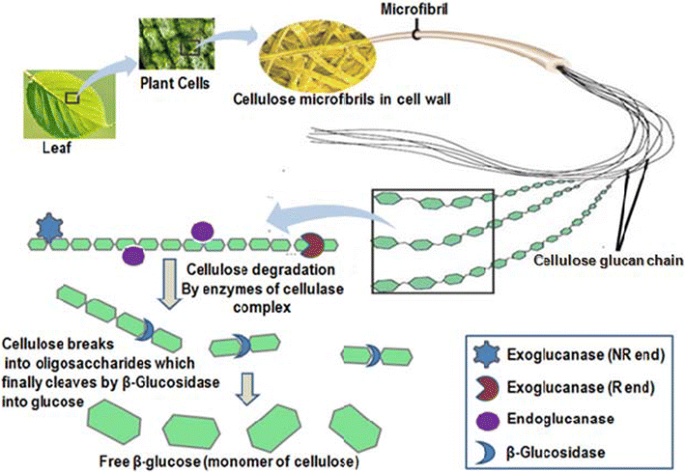
Catalytic properties, functional attributes and industrial applications of β -glucosidases | SpringerLink
PLOS ONE: Several Genes Encoding Enzymes with the Same Activity Are Necessary for Aerobic Fungal Degradation of Cellulose in Nature

Characterization of an extracellular β-glucosidase from Dekkera bruxellensis for resveratrol production - ScienceDirect

Temperature sensitivity of mineral-enzyme interactions on the hydrolysis of cellobiose and indican by β-glucosidase - ScienceDirect
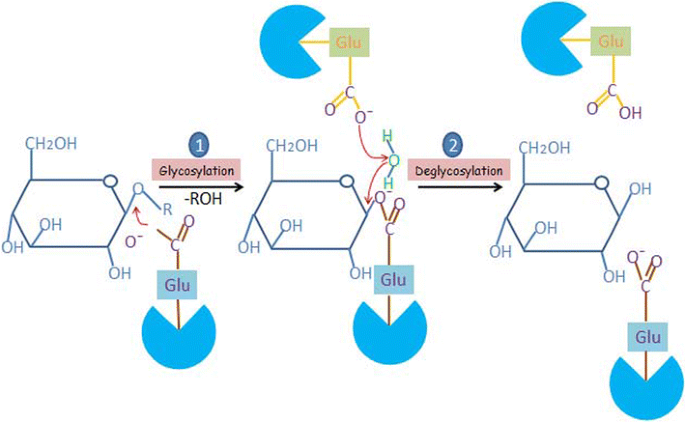
Catalytic properties, functional attributes and industrial applications of β -glucosidases | SpringerLink
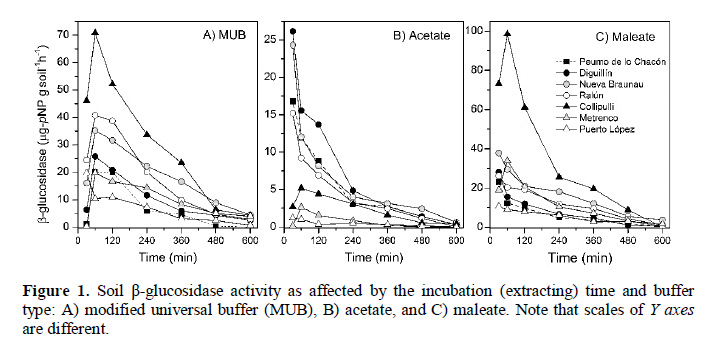
Efficiency of three buffers for extracting β-glucosidase enzyme in different soil orders: Evaluating the role of soil organic matter

A ) Mechanism for an inverting β -glucosidase. ( B ) Mechanism for a... | Download Scientific Diagram